Shohei Kuraoka, Yuhei Higashi, Suguru Saito, Solmaz Pourgonabadi, Honami Honjoh, Sho Ishigaki, Peter C. Harris, Lisa M. Satlin, Michifumi Yamashita, Ryuji Morizane. Deciphering the Impact of RAC1-SPTAN1 in ARPKD Cystogenesis Using Multifaceted Models. Advanced Science, (2026)
doi:https://doi.org/10.1002/advs.202524001
Autosomal recessive polycystic kidney disease (ARPKD) is an inherited renal disorder characterized by fetal-onset kidney enlargement and cyst formation predominantly in the distal nephron. Currently, no established curative therapy exists. In this study, Shohei Kuraoka and colleagues in the Morizane Laboratory employed a multifaceted approach combining a kidney organoid model (organoid-on-chip), transgenic mice, and human kidney tissues. They identified SPTAN1 as a key regulator of cystogenesis in ARPKD. Reduced expression of SPTAN1 led to activation of the RAC1/c-FOS signaling pathway and increased intracellular calcium levels, thereby promoting cyst formation in the distal nephron. Furthermore, restoration of SPTAN1 expression in ARPKD organoid models suppressed cyst formation.
These findings reveal a novel molecular mechanism underlying cystogenesis in ARPKD and suggest that SPTAN1 could serve as a promising therapeutic target, including for epigenome editing-based strategies.
This work was published online in Advanced Science on February 26, 2026.
*Shohei Kuraoka completed his PhD in Kidney development at Institute of Molecular Embryology and Genetics, Kumamoto University under Professor Ryuichi Nishinakamura in 2020. From April 2023 to March 2025, he worked as a postdoctoral fellow in the Nephrology Division at Massachusetts General Hospital under Professor Ryuji Morizane. He is currently working in the Department of Pediatrics at Kumamoto University, as a pediatric nephrologist.
ARPKD is a rare inherited kidney disease primarily caused by mutations in the PKHD1 gene. It is characterized by progressive cyst formation in the distal nephron beginning in fetal life, leading to kidney enlargement, and approximately 10% of patients develop kidney failure within the first year of life. Despite extensive research using PKHD1 mutant mouse models and kidney organoids, the precise mechanisms of cyst formation remain unclear. In particular, recapitulating cyst formation in the distal nephron has been a major challenge in conventional ARPKD model. In 2022, Ryuji Morizane and colleagues established a kidney organoid-on-chip model incorporating fluid flow, successfully recapitulating ARPKD pathology. They demonstrated that aberrant activation of the RAC1/c-FOS pathway is involved in cyst formation (Science Advances, 2022). However, the mechanism of RAC1 activation and the cellular origin of cyst epithelium remained unresolved.
In this study, a multifaceted analysis was conducted using human kidney organoids (organoid-on-chip model), transgenic mice, and kidney tissues from ARPKD patients. Kidney organoids were generated from pluripotent stem cells harboring homozygous PKHD1 mutations. Application of the organoid-on-chip system enabled successful recapitulation of distal nephron-specific cyst formation. RNA-seq analysis revealed that SPTAN1, an interacting partner of RAC1, was significantly downregulated in PKHD1 mutant organoids. Reduced SPTAN1 expression was also confirmed in cyst-lining epithelial cells from ARPKD patient kidneys. Furthermore, organoids derived from pluripotent stem cells with heterozygous SPTAN1 mutations exhibited distal nephron-specific cyst formation and aberrant activation of the RAC1/c-FOS, recapitulating the phenotype observed in PKHD1 mutants. This phenotype was suppressed by a RAC1 inhibitor. Consistently, Sptan1 heterozygous mutant mice also showed distal nephron cyst formation, kidney enlargement, and RAC1 activation. RNA-seq analysis of SPTAN1 mutant organoids further revealed alterations in calcium signaling-related genes, and increased intracellular calcium levels were confirmed in cyst epithelial cells. Finally, restoration of SPTAN1 expression in PKHD1 mutant organoids led to suppression of cyst formation, normalization of RAC1/c-FOS activity, and correction of intracellular calcium levels.
This study demonstrates that downregulation of SPTAN1 triggers aberrant activation of the RAC1/c-FOS pathway, representing a novel molecular mechanism underlying cystogenesis in ARPKD. In addition, single-cell RNA-seq analysis of human kidney tissue and organoid studies suggest that ARPKD cysts may originate from SLC8A1-expressing connecting tubules, located between the distal tubule and collecting duct.
By integrating kidney organoids, animal models, and human tissues, this study provides robust evidence supporting these findings and significantly advances our understanding of ARPKD pathogenesis. Importantly, this is the first report implicating SPTAN1 in cystic kidney disease, highlighting its potential as a novel therapeutic target. These findings are expected to contribute to the development of curative therapies for ARPKD.
ARPKD: Autosomal recessive polycystic kidney disease, a severe pediatric kidney disorder mainly caused by PKHD1 mutations.
Distal nephron: The segment of the nephron that includes from the distal tubule to collecting duct.
Organoid-on-chip model: A system in which kidney organoids are cultured on a microfluidic chip under fluid flow, providing mechanical stimulation.
SPTAN1: A spectrin cytoskeletal protein involved in maintaining cell structure and signal transduction.
RAC1: A small GTPase that regulates cytoskeleton dynamics and cell proliferation.
c-FOS: A transcription factor involved in cell proliferation and stress responses.
SLC8A1: A Na/Ca exchanger important for intracellular calcium regulation.
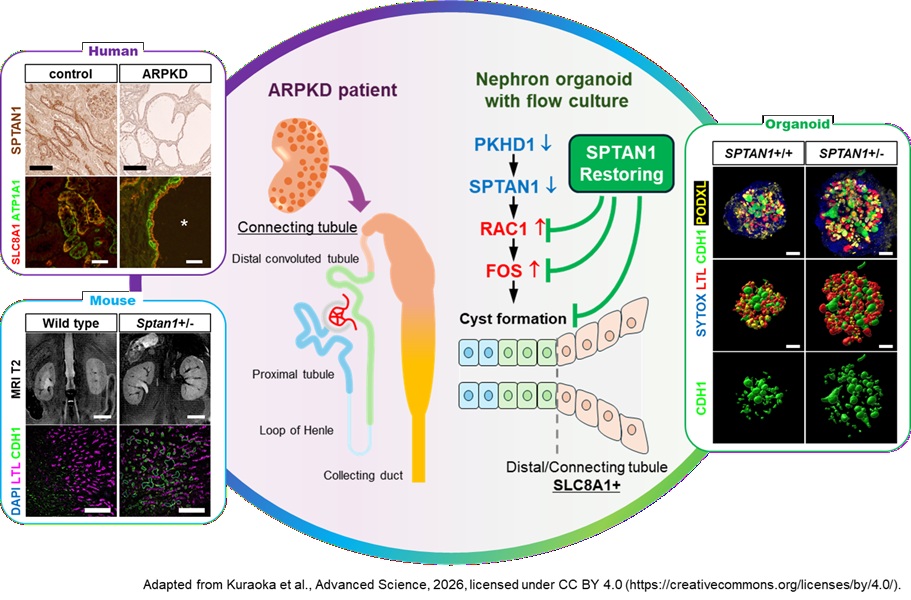
Figure: Multifaceted analysis reveals SPTAN1-dependent RAC1/c-FOS activation and the connecting tubule origin of cysts in ARPKD
PKHD1 mutations lead to downregulation of SPTAN1, which in turn triggers activation of the RAC1/c-FOS signaling pathway, ultimately resulting in cyst formation. In addition, ARPKD cysts are suggested to originate from SLC8A1-expressing connecting tubules, the segment linking the distal tubule and collecting duct. These findings were elucidated through a multifaceted approach integrating kidney organoids, transgenic mouse models, and human kidney tissues.